Reducing enteric methane emissions from ruminants: challenges, solutions and perspectives at the animal and system levels (Full text available in English)
Methane is a greenhouse gas with a strong warming effect on the climate. Scientific knowledge and technologies are evolving rapidly, offering new strategies for reducing enteric methane emissions from ruminants. The consequences of changes in practices and genetic selection objectives must be assessed at the animal, farming system, and territorial levels.
Introduction
The rate of climate change has surged alarmingly. A combination of acute and chronic events, such as heatwaves, temperature peaks, droughts and heavy rainfall, are becoming more frequent and severe. According to the Intergovernmental Panel on Climate Change (IPCC), global warming is projected to reach +2°C by 2050 and +3°C to +4°C by 2100, compared to pre-industrial levels (IPCC, 2023). Despite international environmental treaties, anthropogenic greenhouse gas (GHG) emissions continue to rise, although the rate has been reduced by half over the last decade (IPCC, 2023). Livestock farming accounts for 12% of total anthropogenic GHG emissions (i.e. the emissions linked to human activities). Enteric methane emissions, which are produced when ruminants digest feed, account for almost half of the GHG associated with livestock farming (FAO, 2023). Reducing these emissions is a key climate policy objective, due to the high global warming potential of methane and its relatively short half-life before conversion into carbon dioxide (Box 1). The commitment made at COP26 is to reduce global methane emissions by 30% by 2030 and by 50% by 2050 compared to 2020 levels.
The main sources of methane (CH₄) emissions linked to human activities are livestock farming and rice cultivation (biogenic methane), and fossil fuel extraction, distribution, and landfill sites (fossil methane).
The global warming potential (GWP) of greenhouse gases (GHGs) over 100 years is calculated using the formula GWP100 = CO₂ + 27.8 biogenic CH₄ (or 29.8 fossil CH₄) + 273 N₂O (IPCC, 2023). Biogenic methane is therefore equivalent to approximately 28 times the amount of carbon dioxide (CO₂), which makes its contribution much smaller than that of nitrous oxide (N₂O). However, the half-life of CH₄ is just 10-12 years, making it a key target for governmental policies seeking to rapidly mitigate global warming. In other words, CH₄ has a very strong warming effect in the short term but it does not accumulate in the atmosphere, so reducing methane emissions has a cooling effect.
GWP100 is currently used in all national calculations assessing GHG emissions. New metrics, such as GWP*, are being proposed to better reflect the physics of methane and its short lifetime (Poux et al., 2025). Indeed, GWP* reflects the equivalence between levels of CH₄ and cumulative CO₂ emissions with respect to climate effects, by considering the impact of emission dynamics on the global average temperature over 20 years. Taking GWP* into account in Europe, where CH₄ emissions are declining, would provide a more balanced view of the impacts of CH₄ and N₂O, to define a multi-gas trajectory for the agricultural sector towards 'net zero' CO₂ emissions (i.e. emissions minus carbon sinks).
Europe is one of the few regions in the world where methane emissions have fallen since 1990 (EEA, 2024). The number of ruminants is declining as a result of proactive national policies driven by economic and environmental considerations, such as reducing nitrogen pollution; this decline is also due to dairy cows producing more milk than is demanded, and the difficulties in the renewal of generations of farmers (CGAAER, 2024). This decline is expected to continue over the next 15 years. Reducing the number of cattle alone cannot be the solution, as this would overlook the positive aspects of ruminant farming, including the production of high-quality products and the beneficial role of grasslands and pastures in carbon sequestration and nutrient cycling. Furthermore, an imbalance between the production and consumption of animal products could result in methane emissions being transferred to other countries, providing no benefit at the global level. Consequently, various European countries are expected or have already announced the deployment of farming practices or biotechnologies aimed at reducing enteric methane emissions from ruminants (CGAAER, 2024). The objectives are to make methane emission reductions less dependent on reductions in the number of cattle and the consumption of products derived from them. A number of solutions are proposed, based on feed supplements, ration composition, herd management, and genetic selection. However, climate change affects the availability and quality of plant resources, as well as the ability of animals to adapt to their environment. This may result in inconsistent responses. Additionally, transitions in agricultural and food systems create opportunities, as well as contradictory constraints, for livestock farming and the areas in which it takes place.
Thus, reducing enteric methane emissions from ruminant livestock farming systems in the coming years requires addressing several challenges. The first challenge is to identify optimal strategies that combine existing methods to reduce enteric methane emissions, while ensuring that product quantity and quality, as well as animal health and welfare, are maintained. A second challenge is to identify new levers for reducing enteric methane emissions in a context of increased agricultural system dependence on local conditions, which are themselves affected by climate change. A third challenge is to identify solutions that benefit all aspects of ruminant livestock systems including the impact on other GHG emissions.
This article reviews the studies conducted by INRAE and its research and development partners over the past decade, with the aim of reducing methane emissions from ruminants. Enteric methane is produced when microorganisms (methanogenic Archaea) break down feed (forage and concentrates) into volatile fatty acids (VFAs), using hydrogen for their metabolism and growth (Figure 1).
Within the rumen, the pathways involved in hydrogen (H₂) consumption are represented by arrows between compartments, with minimal intensity for microbial biomass, average intensity for propionate production (VFA, volatile fatty acids) and maximum intensity for methane (CH₄) production. The scientific priorities, which are supported by a better understanding of the interaction between the rumen and the host, are shown in boxes on the right and left of the figure, respectively.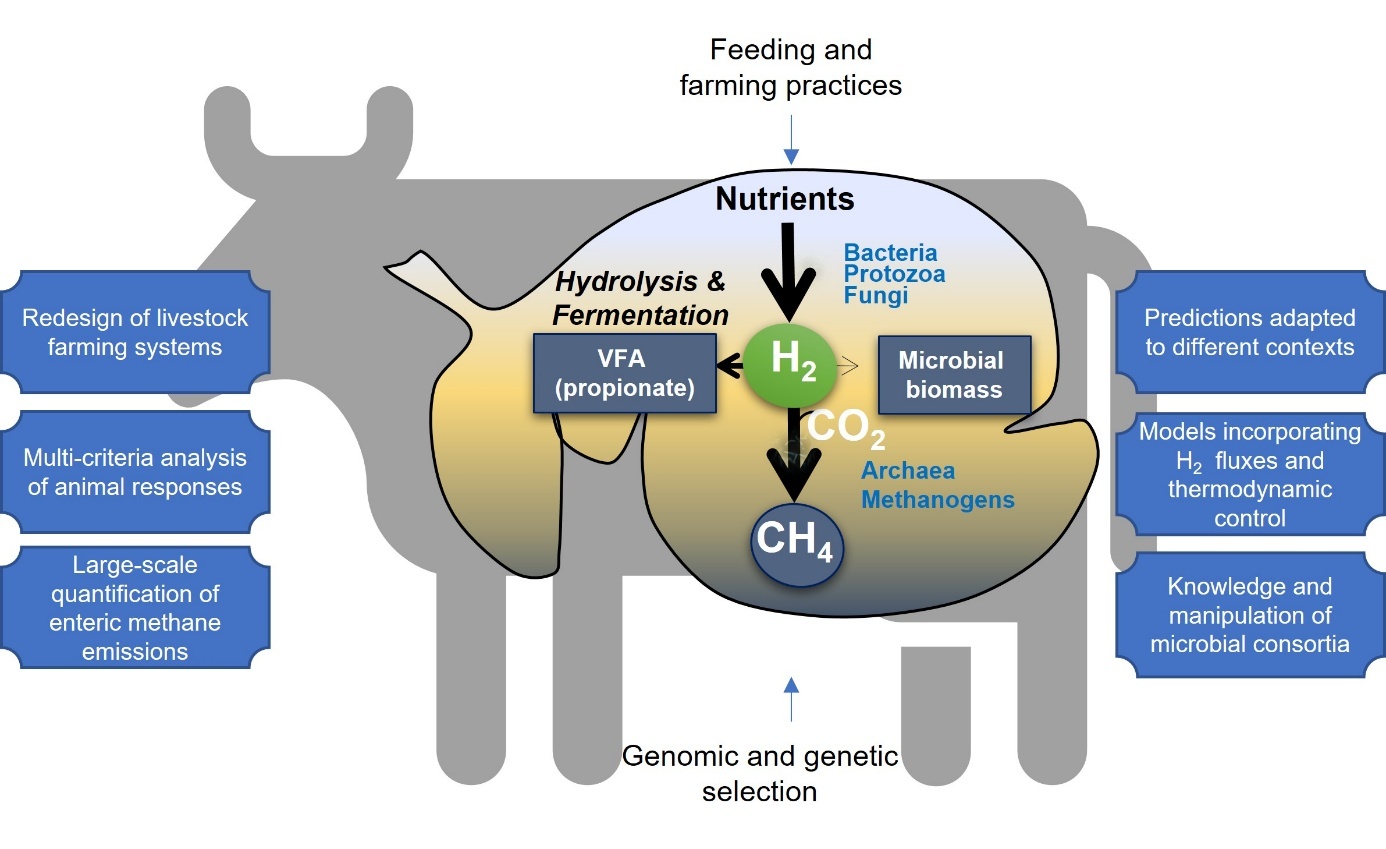
Figure 1. Components of enteric methane emissions and research objectives.
To assess the effectiveness of different levers, accurate tools are required to measure and evaluate enteric methane emissions in various contexts (Part 1). To identify new targets, it is necessary to characterise and understand the microbial biomass diversity and the microbiome functions at key stages in the animal’s life (Part 2). Strategies to reduce enteric methane emissions include genetic selection (Part 3) and feeding strategies and farming practices (Part 4). However, it is important to consider their impact on animal performance and farming systems. To promote changes in farming systems, we require a better understanding of how these practices can be implemented in both temperate and hot environments with a gradient of pedoclimatic constraints (Part 5).
1. Tools for phenotyping enteric methane emissions
Research programmes use a set of direct and indirect measures, aiming to advance our understanding of the biological mechanisms underlying methane emissions and to develop tools adapted to on-farm constraints. Direct measurement methods quantify methane emission fluxes, whereas indirect estimation methods rely on prediction models. The predictive variables may relate to the animal’s rations and/or production characteristics, such as live weight and productivity, among others. They may also be derived from the analysis of various biological samples taken from the animal, including emitted gases, milk, blood or faeces.
1.1. Direct measurements to acquire reference data
Various devices have been developed that enable direct measurements of enteric methane emissions. These devices are now used by most research organisations (Mesgaran et al., 2021), particularly in INRAE’s experimental units. These include respiratory chambers, which are considered the reference method, and sulphur hexafluoride (SF6), a tracer gas that is administered orally with a vector which allows for controlled, constant release into the animal’s rumen. Another device is GreenFeed®, which takes spot measurements of the methane emitted by animals when they visit an integrated automatic concentrate dispenser. Doreau et al. (2018) showed comparable average methane emission levels between the three measurement devices for groups of animals in European livestock systems and validated the reliability of GreenFeed® measurements on-farm. However, the workload involved in using GreenFeed®, the cost of acquiring and operating the device and the reliance on the company for data collection are major obstacles to its use on commercial farms. The applicability of these devices in tropical environments has also recently been discussed (Gbenou et al., 2024a).
1.2. Indirect measures for assessing large numbers of animals
For several years, equations have been developed to predict enteric methane emissions, by establishing the relationships between measured emissions and various explanatory factors such as feed intake and diet composition, for different categories of animals (e.g. INRA, 2018; van Lingen et al., 2019; Belanche et al., 2023). Measurements in biological matrices obtained from the animals (milk and faeces) are also used for larger-scale estimates. Initially, the fatty acid (FA) composition of milk was used as an indicator of methane emissions in dairy cows fed maize silage containing extruded linseed (Chilliard et al., 2009), and subsequently in cows fed a wide range of diets (Bougouin et al., 2019). Determining the FA composition by gas chromatography is expensive and time-consuming. Therefore, equations based on the detailed FA composition are not suitable for measuring large numbers of animals. Conversely, the mid-infrared (MIR) absorption spectra of milk are now routinely collected for millions of cows to predict the milk composition for dairy inspection purposes. These spectra have been used to predict methane emissions in dairy cattle (Vanlierde et al., 2021; Fresco et al., 2024b) and enable genomic evaluation to select low emitted dairy cattle (Part 3). Prediction equations have also been developed using near-infrared absorption spectra (NIRS) acquired from faeces, a preferred matrix for suckler herds and for growing dairy animals. Promising results have been obtained using a dataset of suckler cows for which methane emissions were measured using the GreenFeed® method (Andueza et al., 2022).
However, the robustness of the various prediction equations must be assessed to accurately estimate the reductions that can be achieved by mitigation strategies at the level of the livestock farming systems (Benaouda et al., 2019; Blondiaux et al., 2024).
1.3. Prospects for developing measurement tools or predictors
Direct and indirect measurement tools should complement one another. This is essential for obtaining accurate predictors. The experimental infrastructures of INRAE and its research and development partners are acquiring new devices to complement those already in use (Box 2). These tools will be essential for future studies aimed at testing the most effective strategies for reducing enteric methane emissions in various farming environments. Portable Accumulation Chambers (PACs) are now available for small ruminants. They measure the gases emitted by flatulence over a period of 1 hour, as well as the methane emitted by belching and exhalation (Goopy et al., 2011). Sniffers are usually located inside milking robots and use infrared technology to measure the animals’ methane emissions. The predictive potential of these indirect methods must be compared with the predictions based on MIR spectra in milk. Developing predictors adapted to farm constraints is indeed a priority. In France, sniffers will soon be installed on 50 commercial dairy farms, and PAC will be used on various commercial small ruminant farms to collect data. This will enable comparisons to be made between farming systems and the predictors to be evaluated in different contexts.
The conditions and procedures for quantifying or estimating enteric methane emissions from ruminants using different methods have been documented in a methodological publication by SmartCow (Mesgaran et al., 2021). This European consortium is coordinated by INRAE and brings together experimental cattle infrastructures.
The tools that implement these methods are now in use at INRAE’s experimental infrastructures across France. These include Le Pin in Normandy (GreenFeed® and sniffers) and the Experimental Dairy Production Facility of UMR Pegase in Brittany (GreenFeed®) for dairy cattle; Herbipôle in Auvergne for dairy or suckler cattle and sheep (GreenFeed® and respiratory chambers); the Small Ruminant Phenotyping Centre in the Loire Valley (GreenFeed® and sniffers) for sheep; and the Tropical Livestock Platform for Agroecology (PTEA) in Guadeloupe (GreenFeed® and sniffers). Climate-controlled metabolic chambers for cattle and Portable Accumulation Chambers (PACs) for small ruminants will soon be available in experimental stations, including Herbipole, the Small Ruminant Phenotyping Centre, the La Fage experimental unit in Aveyron and the experimental facility of GenPhySE in Occitanie.
The Institut de l'Elevage (Idele) has recently acquired several GreenFeed® units and sniffers. These can be used to conduct experiments with dairy cattle, beef cattle and small ruminants.
Improving the accuracy of already existing predictors is also essential for extending their use. Complementary research studies in cattle and small ruminants will be conducted to evaluate the potential of milk MIR spectra or faecal short-wave infrared (SPIR) spectroscopy to predict methane emissions. Other new predictors are under study, including plasma metabolites such as amino acids, glucose, carboxylic acids and glycerolipids (Yanibada et al., 2020), the carbon isotopic abundance of milk (Saro et al., 2025), microorganisms and hydrogen fluxes (Bedoya Mazo et al., 2023).
Continuous data acquisition, supported by animal experimentation under national and international collaborations, remains essential. Combining the databases will enable us to develop more robust predictors and discriminating models of methane enteric emissions. It will also ensure that inter-individual variability is considered when evaluating farming systems in different pedoclimatic contexts (Part 5).
2. Rumen microbiota: modulating hydrogen fate and limiting methane production
The rumen microbiota is a key focus of strategies aimed at reducing enteric methane emissions. Indeed, methane is produced through the degradation and fermentation of feed by rumen microorganisms. Archaea, a branch of microorganisms, produce methane through a series of reactions known as methanogenesis. There are several pathways of methanogenesis, but the most common in the rumen involves the reduction of carbon dioxide by hydrogen. Archaea do not produce these two compounds, but other microorganisms such as bacteria, protozoa and fungi do (Morgavi et al., 2010). As methane production is a natural consequence of the metabolic processes of the microbiota when producing energy substrates (VFAs) for the host, any strategies aimed at reducing enteric methane emissions must not have a negative impact on the supply of VFAs. This dual challenge has been addressed.
2.1. Hydrogen transactions in the rumen microbiota
Manipulating the production and consumption of hydrogen is considered a key approach to mitigating enteric methane emissions (Martin et al., 2010; Mackie et al., 2024). Researchers have focused on evaluating alternative hydrogen acceptors, to divert hydrogen away from methanogenesis. Guyader et al. (2017) demonstrated that supplementing diet with nitrates, an oxidant that can be reduced by hydrogen, can stimulate metabolic pathways that accept hydrogen, thereby competing with methanogenesis. However, the underlying microbial processes remain to be elucidated. Other alternative hydrogen acceptors such as phenolic compounds have been studied using in vitro approaches (Huang et al., 2023; Romero et al., 2023). Phenolic compounds are secondary plant metabolites that can be metabolised by microbial populations into acetate, while consuming hydrogen. Of the 10 or so phenolic compounds tested at INRAE, gallic acid and phloroglucinol (which were derived here from the chemical industry, but are also found naturally in tree bark) increased gas production and total VFA production, particularly acetate, without stimulating methane production. The effect of phloroglucinol was evaluated in combination with 2-bromoethanesulfonate, a specific methanogen inhibitor. Methane production was completely inhibited, hydrogen accumulation was reduced and acetate production increased compared to treatment with 2-bromoethanesulfonate alone. Research programmes on hydrogen transactions are on-going at INRAE, with a particular focus on quantitative data within the rumen ecosystem.
2.2. Strategies for young animals
After birth, the rumen is colonised sequentially by microbiota. Microorganisms are present from the second day of life and are characterised by the abundance of Pseudomonadota (formerly Proteobacteria) which accounts for nearly 70% of the total. The composition changes rapidly, with the proportion of Pseudomonadota falling to 30% before the ingestion of solid food. Meanwhile, the proportion of Bacteroidota, particularly Prevotella, increases to over 50%. If any induced changes in rumen microbiota persist in the long term, the plasticity of this microbiota at the beginning of an animal's life may provide opportunities to reduce enteric methane emissions. In dairy calves, administering 3-nitrooxypropanol (3-NOP), a methanogenesis-inhibiting additive, from birth until 14 weeks of age resulted in a lasting reduction in enteric methane production, which was still observable at 1 year of age (Meale et al., 2021). The composition of bacteria, and to a lesser extent that of Archaea, was altered in the rumen, and this alteration persisted with some variations even after the administration of 3-NOP ceased. Similarly, by supplementing the diet of pregnant heifers - and then, their calves from birth to weaning - with 3-NOP, Martinez-Fernandez et al. (2024) reported a persistent reduction in methane emissions 28 weeks after stopping the treatment for at least one group of treated calves. The rumen microbiota of these calves was similar to that of the low-emitting adult cows found in Prim'Holstein INRAE herds (Ramayo-Caldas et al., 2020).
Such early modulation of the microbiota enables constraints associated with different cattle management practices, such as grazing, to be overcome. Indeed, when grass is mature and not particularly diverse, grazing increases methane emissions in ruminants compared to a diet based on energy-rich concentrates (Roques et al., 2024).
2.3. Contribution of mechanistic modelling
Mechanistic modelling is a complementary approach to animal experimentation for representing and predicting rumen metabolism. Muñoz-Tamayo et al. (2016) proposed a model of the rumen microbiota organised into functional groups according to their utilisation of glucose (hexoses), amino acids, and hydrogen. This model includes glucose utilisation flux allocation factors and hydrogen dynamics. It is crucial to consider hydrogen in the model. However, our understanding of the microbial processes and thermodynamic rules that govern hydrogen transactions is incomplete, particularly for quantifying hydrogen fate and the impact of energy allocation on microbial fermentation (Morgavi et al., 2023). Nevertheless, this model can accurately depict in vitro fermentation data from mixed diets containing different levels of concentrates (Serment et al., 2016). It does not consider biological processes, such as VFA absorption rates and the passage of substrates. This model is currently being extended to incorporate these in vivo conditions, and preliminary results comparing simulated and experimental data from a small number of Nordic Red dairy cows are promising (Muñoz-Tamayo et al., 2023a). In parallel, a modelling approach has been developed that links microbial data to VFA dynamics; this uses a semi-continuous in vitro system (RUSITEC) that mimics in vivo conditions, and data obtained from the rumen of dairy cows (Davoudkhani et al., 2024).
The model developed by Muñoz-Tamayo et al. (2016) was also expanded to represent the effect of the macroalga Asparagopsis taxiformis on the dynamic profile of microbial fermentation measured in vitro. The inhibition of methane production by this macroalga leads to the accumulation of hydrogen, which exerts thermodynamic control over fermentation by directing the flux towards VFA production. The models proposed by INRAE (Muñoz-Tamayo et al., 2021) and van Lingen et al. (2021) are the only two mechanistic models capable of representing the impact of anti-methanogenic additives. At the microbial level, modelling kinetics enabled the quantification of metabolic and energetic differences between three methanogen species (Muñoz-Tamayo et al., 2019). The data suggest that, in addition to kinetic and thermodynamic factors, other forces such as microbial interactions (e.g. endosymbiosis with protozoa and microbial aggregation) and spatial variation (e.g. fractions of liquid and particles) contribute to the ecology of the methanogen community in the rumen, thereby promoting microbial diversity.
Integrating genomic information from the microbiota should improve the predictive accuracy of the models (Muñoz-Tamayo et al., 2023b). The genomic sequence of the cellulolytic bacterium Fibrobacter succinogenes S85 can be used to reconstruct its metabolic network. Then, it is possible to develop a dynamic metabolic model that predicts the production of acetate, succinate and formate from glucose, cellobiose and cellulose metabolism (Fakih et al., 2023). This approach will soon be extended to other rumen microorganisms, including methanogens.
2.4. Research prospects for microbiota
A better understanding of how microbiota produce methane could lead to the development of new strategies reducing enteric methane emissions, by managing rumen hydrogen metabolism, modulating the microbiota during early life and using mechanistic modelling to address microbial complexity. This would help identify the trade-offs between reducing methane emissions, improving feed efficiency and production performance, and ensuring animal health and welfare.
Two main research perspectives have been identified to achieve this:
(i) Elucidating the dynamics of the rumen microbial ecosystem, particularly its resilience, and the interactions between microbial species. This would improve our understanding of the rumen microbiota and allow us to consider this dynamic more effectively in future strategies to reduce enteric methane emissions.
(ii) Develop a continuum of in vivo, in vitro and in silico studies. In vitro approaches enable the fermentation pathways associated with substrates and additives, as well as the related physicochemical parameters, to be evaluated. This simplifies the complexity observed in vivo. In silico approaches are based on in vitro and in vivo methods to predict the effectiveness of proposed strategies. The continuum operates in both directions: towards in silico for understanding and towards in vivo for application. This synergy in elucidating microbial mechanisms and proxies contributes to the principles of the 3Rs - replace, reduce, and refine - of using animals for scientific purposes.
3. Genetic levers for reducing enteric methane emissions
For a trait to be selectable, it must be measurable on a relatively large scale, and there must also be genetic variability between individuals. The first condition is quite limiting because it is often difficult to measure methane emissions outside of an experimental infrastructure (i.e. for more than a few hundred animals). Although relatively new measurement devices, such as sniffers, could be helpful, they are not yet fully operational in France. Therefore, two main options have been chosen to reduce methane emissions through genetics.
3.1. Selection against methane emissions predicted from milk MIR spectra
Predicting methane emissions from MIR spectra enables large populations of phenotyped animals to be obtained. Indeed, MIR spectra are acquired in very large numbers, approximately 15 million per year from around 2.5 million dairy cows, and the marginal cost of predictions is low once the equations have been established. Two research and development consortia (Optimir and MethaBreed) have developed equations based on reference measurements from respiratory chambers or using the SF6 method (Fresco et al., 2021), or by using GreenFeed® in nine experimental conditions (Fresco et al., 2024b). These equations can be used to predict various emission traits, such as per animal (g/day), per production unit (g/kg standardised milk) or per intake unit (g/kg dry matter intake). Daily methane production can also be calculated by multiplying methane emissions expressed as per kilogram milk, by the daily quantity of milk produced. These equations have moderate accuracy. This shortcoming is partially offset by the large amount of data, which significantly reduces loss of accuracy at the genetic level without eliminating bias.
Fresco et al. (2024a) subjected predictions to an in-depth genetic analysis, which showed that heritability is quite strong. This is not surprising, given that the absorbances of the most informative wavelengths in the MIR spectra are relatively heritable themselves. The genetic correlations between predicted traits are moderate, with the highest correlation (0.6) found between methane emissions in gram per day predicted by the two equations established by the two consortia. This moderate correlation can be explained by the fact that the two consortia used different measurement methods and calibration populations. With the exception of the reconstructed prediction, which is closely linked to milk yield, the correlations with production traits are moderate. Moreover, the reconstructed prediction is challenging to utilise, as a decrease in enteric methane emissions would result in selection against milk production.
The pilot genomic evaluations are available (Fresco et al., 2024c). They were carried out using a single-step model, based on predictions from the official milk recording of several million cows, out of which several hundred thousand were genotyped. This method simultaneously analyses the performance of both genotyped and untyped animals. It implicitly estimates the genotype expectation of untyped animals based on the genotyping of their relatives. This increases accuracy and reduces bias. These genomic evaluations demonstrate the potential of young bulls in reducing enteric methane emissions. Furthermore, they demonstrate that the genetic potential of animals for methane emissions has been minimally impacted by recent selection objectives.
Genomic evaluations are expected to be officially launched by selection organisations in 2026. The methane trait (g/day) is the average of the two equation predictions established by the two consortia. This is a more robust approach than selecting just one of the two predictions when there is a lack of objective criteria on which method is superior. In the absence of direct economic value for the farmers, the weight given to methane emission reduction in the overall selection objective, as defined by selection organisations, may be based on the expected gain. Targeting 1% annual reduction in methane emissions (in g/day) means a weight of around 25% for the methane trait in the selection objective (Fresco et al., 2025). This would moderately slow down the genetic progress for the other traits already under selection. Although no adverse effects have been observed to date, it will be necessary to confirm that selecting against methane does not affect the digestive processes of the ruminants. It is not feasible to use milk MIR spectra in suckler cows, as they are not milked. An approach based on SPIR spectra in faeces is currently under study. If this proves effective, then a measurement system will need to be developed before selection against methane emissions in suckler cows can be considered.
3.2. Improvement of breeding traits correlated with methane emissions
Predictions based on milk MIR spectra are moderately accurate. Many factors influencing methane emissions are not, or are only partially, considered in MIR predictions based on milk production. Therefore, to reduce methane emissions more effectively, it is also appropriate to address these factors in addition to direct genetic selection. Most of these factors depend on both farming systems and genetics. Therefore, the content presented in this section is relevant to the implementation of changes to farming systems. Improvements in breeding traits correlated with methane emissions, such as age at calving, fertility, health and body size, generally have a positive effect on the economic performance (reducing feed and veterinary costs) of the farms.
The key solutions are reducing losses, particularly mortality, and minimising unproductive periods, primarily by lowering the number of heifers.
One option is to reduce the age at first calving. In France, the age at first calving ranges from 2 to 3.5 years; more than 65% of the dairy and suckler herds calve at 3 years of age or older. Reducing this age of first calving at 2 years would save an annual cohort of 1 million near-adult-sized animals, each producing 250–300 g of methane per day. This would primarily affect the farming system, but selecting for precocity could also be helpful, especially for suckler breeds which tend to mature later. Although calving at 2 years of age may seem ambitious, any progress towards greater precocity is desirable despite the various obstacles. For suckler cows, it is also important to minimise the number of years without calving, which implies maximising fertility.
A second option is reducing the replacement rate. Although genetics is not the most important factor, improving longevity by selection is possible, with gains of +0.5 to +1.0 lactation being achieved. This would decrease the need for replacement heifers. Reducing the replacement rate by 25% per year would contribute to a 6% reduction in methane emissions from French dairy cattle with a first calving at two years of age. With first calving at three years of age, the reduction in methane emissions would be 10%. These results are illustrated by Dall-Orsoletta et al. (2019). Reducing the rate of involuntary culling would also lower the replacement rate. This could be achieved by improving health and fertility. Health problems result in production losses and unnecessary methane emissions. It is now possible to select for health, with multiple indicators available such as udder health, leg health, and susceptibility to diseases (paratuberculosis and ketosis). Thus, health and fertility are important traits in dairy selection objectives.
A third option is to reduce methane emissions related to maintenance requirements. French dairy and suckler cattle breeds are characterised by their large size. The bigger the animal, the more feed it consumes and the more methane it produces during the breeding period. Reducing adult weight by 100 kg is possible by selection. This would reduce maintenance requirements by 10%, and lead to a 3% to 5% decrease in methane emissions (depending on the relative importance of maintenance in total requirements). This would also have favourable effects on other traits, such as precocity and longevity. Selecting for a smaller size could be achieved without adversely affecting milk production, as there is only a very moderate genetic correlation between the two traits. Precocity in animals reared for meat, which is facilitated by the reduction in adult size, would also favour fat deposition; this would reduce the age at which the animals are slaughtered and the amount of concentrates needed.
Finally, in a given farming system, improving animal productivity is widely used as a GHG mitigation strategy (Dall-Orsoletta et al., 2019): low-producing dairy cows produce more methane per kilogram of milk, and thus, should be avoided. Selecting animals with better feed efficiency and improved robustness (i.e. better adapted to the environment) is also an option.
3.3. Prospects in genetics
There are many genetic options to reduce methane emissions, both directly and indirectly. One strategy, such as in the on-going project Methane 2030 (Box 3), is to construct a synthetic methane index that incorporates all traits (direct and indirect) associated with variations in methane emissions. The success will depend primarily on the selection pressure – that is, how much weight is given to methane in the selection objective. Farmers must be financially incentivised for their efforts before they agree on high selection pressure. This means that progress in reducing methane emissions can be measured and valued economically on markets. Therefore, voluntary public policies and/or industry initiatives are required. To reduce methane emissions by 1% per year, the selection objective must place a higher value on methane than the current market price of carbon. The value must reflect the situation in 8-10 years from now, since the effects of genetic selection only become apparent in the long term.
4. Dietary and management solutions for reducing enteric methane emissions
Enteric methane emissions are associated with microorganisms in the rumen, but also to the quantity of ingested organic matter that is fermented, and depends on ruminant’s physiology and genetics. Thanks to scientific progress, a large number of levers are now available, but they are at different technology readiness levels (TRLs), ranging from scientific discovery to large-scale implementation on commercial farms (Table 1). Some countries are also developing vaccines based on genomic information from rumen methanogens, but the potential effects on methane emissions are still unknown.
Compared to additives, which are the most effective strategies for decreasing methane emissions by 20–40%, dietary strategies have modest effects, reducing emissions by –10% and –20%. However, they often have fewer negative effects on other traits at the animal or farming system levels (Table 1). Unlike genetic factors, dietary strategies and additives have an immediate impact on reducing enteric methane emissions. The observed reductions associated with dietary strategies vary, depending on the concentrations of specific active plant components, the interaction effects with other feed compounds, the physiological stage of the animal, and/or other confounding factors.
The decrease in enteric methane emissions associated with each lever also depends on the mode of expression: “absolute” emissions (in g/day) per animal, “emissions yield” per unit of intake (in g/kg of dry matter intake), or “emissions intensity” per unit of product (in g/kg of standardised milk or meat). Absolute emissions (g/day) are better suited to public policy and expected climate benefits, so they should be prioritised. Other modes of expression (e.g. g/kg of product) address the specific challenges faced by farmers and industries more directly. This could lead to conflicts between stakeholders regarding acceptance of the proposed solutions.
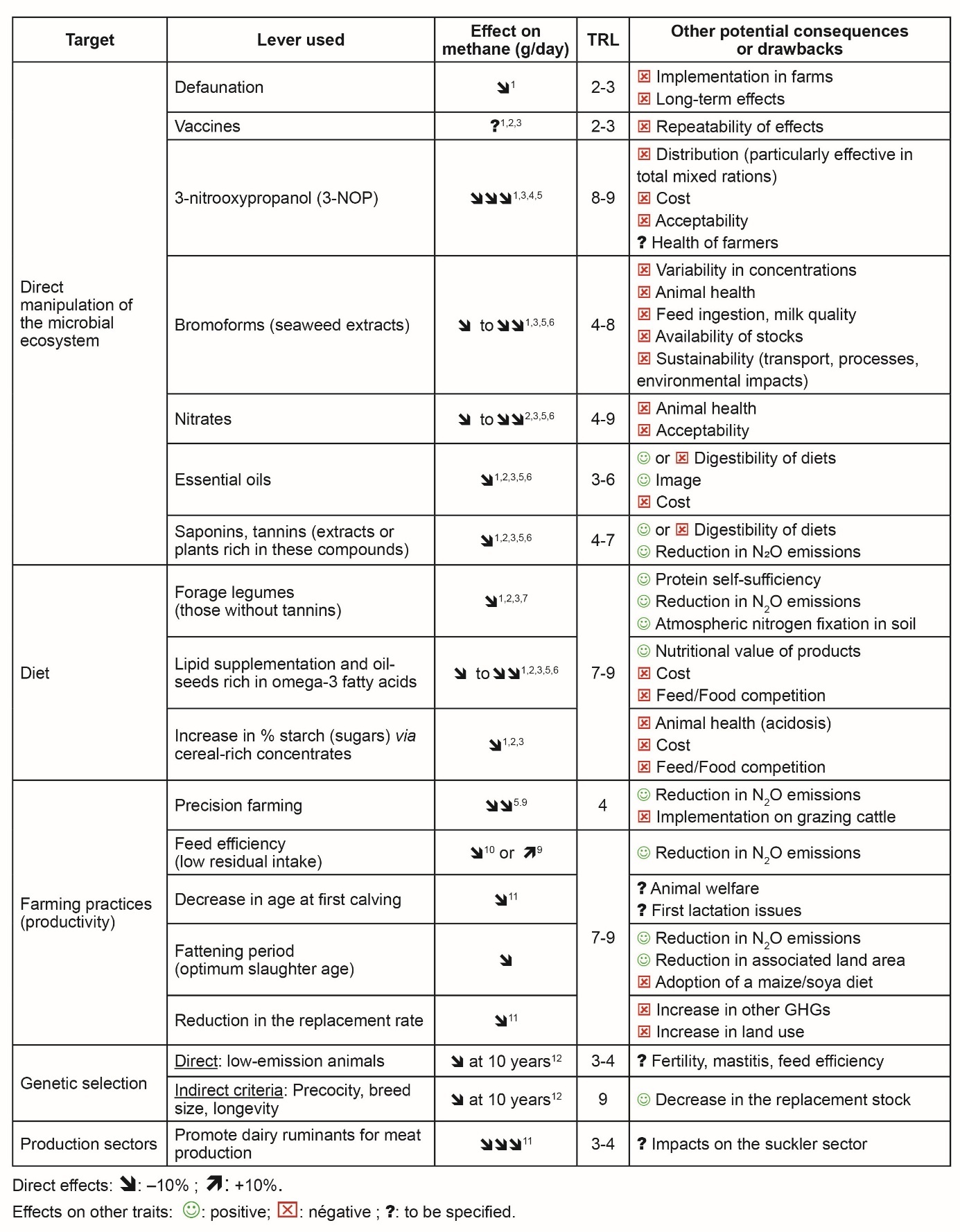
According to 1 Beauchemin et al. (2022); 2 Martin et al. (2010); 3 Roques et al. (2024); 4 Morgavi et al. (2023); 5 Martins et al. (2024); 6 Martin et al. (2021); 7 Archimède et al. (2011); 8 Niderkorn & Jayanegara (2025); 9 Fischer et al. (2020); 10 Bes et al. (2022); 11 Dall-Orsoletta et al. (2019) and 12 de Haas et al. (2021).
TRL (Technology Readiness Level): 1 – basic principles – to 9 – proven in operational environment.
5. Enteric methane emissions for farming systems located in contrasting pedoclimatic areas
5.1. Variability in enteric methane emissions at the system level
Livestock farming systems include different management practices in both temperate zones and under hot climates, such as free-range, grazing, rangeland, agroforestry and mixed rations combining fodder and concentrates, among others. There are also various types of feeds, including co-products, crop residues, industrial by-products, and tree and forage species. This results in a wide range of enteric methane emissions between systems. Notably, enteric methane emissions vary between temperate and tropical zones (Archimède et al., 2011; Martin et al., 2021; Gbenou et al., 2024b). The types of feed used differ, as do the breeds. A better understanding of the physiology of some breeds is still needed.
Feeding systems provide valuable insights into feed composition, animal characteristics and feeding practices in temperate regions. For example, the latest version of INRA feeding system (INRA, 2018) calculates methane emissions based on digestible organic matter, considering feed intake and the proportion of concentrates in the ration. It estimates a decrease in methane yield for each percentage increase in concentrates. However, feeding systems are less suitable for ruminant farming systems in hot areas. A literature review by Quantin et al. (2025) indicates that the methane emissions per kilogram of dry matter intake are 23.2 (range 16.9–29.1) for dairy cows and 27.8 (range 18.7–34.5) for heifers in sub-Saharan Africa, compared with an average of 21.0 in temperate zones. The values are 21.9 (range 16.8–26.5) and 31.7 (range 12.6–49.8) for sheep and goats, respectively, in sub-Saharan Africa, compared with an average of 18.0 in temperate zones.
Equations can be used to predict methane emissions and nitrogen excretion to quantify the emissions of all pollutants associated with livestock farming. However, they are subject to uncertainty (Benaouda et al., 2019) and are not always applicable to plants that are rich in secondary bioactive compounds or to additives that have specific modes of action. Emission factor uncertainty and lack of metadata result in the inadequate consideration of the environmental impacts of livestock in GHG calculators and inventories (Eugène et al., 2019; IPCC, 2019). Nevertheless, these issues can be addressed by dedicated methods, to improve the selection of instruments for the direct measurement of GHG emissions (Hammond et al., 2016), as well as by relevant proxies (Vanlierde et al., 2024) and models (Blondiaux et al., 2024).
5.2. Redesigning farming systems to reduce enteric methane and other GHG emissions
At the farming-system level, strategies to mitigate enteric methane emissions often involve increasing herd productivity, longevity and reproduction. The aim is to reduce emissions through dilution while maintaining economic viability. In temperate regions, highly specialised livestock farming systems that prioritise technical and economic performance can result in lower methane emissions per unit of milk or meat produced. However, these systems produce more methane per unit of surface area due to the high concentration of animals, and result in significant environmental damage through high nitrogen emissions into the air and water, as well as reduced biodiversity (Chemineau et al., 2025). Conversely, agroecological systems increase soil carbon storage, promote biodiversity and reduce nitrous oxide emissions. However, they have little effect on methane emissions (Blaix et al., 2026). Strategies to reduce enteric methane emissions could be introduced, provided they are compatible with agroecological principles that promote interactions between organisms and their integration into the local ecosystem.
Numerous studies have highlighted the benefits of mixed farming systems, which improve environmental efficiency by incorporating agroecological principles, such as closing nutrient cycles and promoting plant and animal biodiversity. In hot regions, other strategies include improving pasture quality, supplementing diets, and changing land use and ruminant breeds (Thornton & Herrero, 2010). Natural grasslands are often used for grazing livestock. Farmers deal with the lack of feed by managing rations every day (e.g. using woody and herbaceous forages) and by moving livestock to new pastures (transhumance). In these systems, there are large seasonal variations of fodder quality and availability. Studies in the Sahel (Assouma et al., 2018; Bois et al., 2020) have assessed the impact of this seasonality, demonstrating that fodder intake during the dry season results in increased methane production in vitro. Supplements can reduce methane emissions per kilogram of dry matter ingested, particularly when nitrogen intake is not limiting (Gbenou et al., 2024b).
Agropastoral farming systems are examples of agroecological systems because they exploit the complementarity and synergy between spontaneous and cultivated plants and animal diversity (behaviour and performance). They generally have low environmental impacts (Vigan et al., 2017). Although they are more vulnerable to hazards, they offer better adaptability and mitigation capacity by promoting mixed farming (or even poly-livestock farming), which has positive effects on carbon storage and manure utilisation. Analysing nutrient flows provides a better understanding of efficient processes and those associated with methane emissions (Puech & Stark, 2023). This allows researchers to propose options that promote synergies between carbon and nitrogen. Indeed, livestock play a crucial role in the spatial distribution of organic matter, soil nutrients and carbon stocks. For example, in tropical ecosystems, around 50% of the dry matter in fodder is returned to the soil (Schlecht et al., 2004). The carbon balance of a pastoral area can even be negative when all emissions are considered. In a semi-arid silvo-pastoral landscape (Assouma et al., 2019), the amount of carbon fixed over the annual cycle (in soil organic matter, wood, and tree roots) exceeds the emissions caused by animals and their manure (methane fermentation from livestock and termites, GHG emissions from soil and water, and vegetation fires and motorised pumps).
Together, polyculture-livestock systems in harsh environments can provide inspiration for mitigation strategies in temperate regions, particularly those most susceptible to climate change.
5.3 Prospects for research and development on farming systems
The first challenge is to improve our understanding of, and develop new tools for managing, animal and plant diversity in different regions and climates. This involves calibrating existing models using specific data sets and/or developing context-specific models based on the local animal and plant resources.
A second challenge is developing appropriate indicators or metrics with which to assess the contribution of different livestock systems to sustainability. In this context, it is important to consider methane and other GHG emissions together with carbon storage and nutrient cycling (nitrogen and phosphorus).
Conclusion
Thanks to methodological advances in measuring methane emissions from ruminants, particularly MIR spectroscopy of milk and SPIR spectroscopy of faeces, it is now possible to evaluate large populations of animals under a variety of conditions. These methods facilitate evaluation of the benefits of methane emission reduction strategies in a variety of farming contexts. This is important for their application in different regions which depends on factors such as the availability of feed resources, geography and vulnerability to climate change. These developments, combined with a better understanding of biological processes, have also made it possible to create prototypes of selection models for low-emitting ruminants. However, genetic progress, including genomic selection, takes time – at least 5 years. Due to the urgent need to mitigate global warming, it is important to modify the selection objectives for various breeds by combining direct and indirect genetic approaches aimed at reducing enteric methane emissions.
Research programmes aimed at identifying the biological mechanisms underlying methane emissions in animals must continue, particularly those dedicated to studying the relationship between microbial diversity in the rumen and host phenotypes. High-throughput methods are now available for defining syntrophic relationships between microorganisms. Methods for multimodal data integration are also available, which will improve predictive models and multi-criteria evaluation. This will enable the identification of new solutions based on a better understanding of hydrogen transactions and electron capture and/or the promotion of specific microbial consortia, while maintaining the ability to digest cellulose. This will ensure production performance, health and welfare of the animals.
Feed resources are an important component of farming systems that can help reduce methane emissions. Various forages adapted to climate change, legumes (particularly tannin-rich varieties) and co-products could be incorporated more systematically into ruminant rations. This requires knowledge of the nutritional value of different plants and their effect on GHG emissions (methane and nitrous oxide). It also requires a better understanding of how to adapt to variability in their composition under global warming and how to consider their availability depending on the pedoclimatic context and competition between feed, food and fuel in the region. Thanks to the progress that has already been made, as well as the ongoing research and development projects (Box 3), it will be possible to integrate the nutritional value of plant resources for milk or meat production and their environmental impacts into decision-support tools. Stakeholders will then decide on the best compromises, taking spatial (i.e. complementarity within the territory) and temporal (i.e. across seasons) issues into account.
The Methane 2030 project was launched in 2024 for 4 years with the aim of achieving a 30% reduction in enteric methane emissions in France within 10 years.
The objectives are (i) to harmonise data acquisition systems for large-scale deployment; (ii) to develop methane emission prediction methods adapted to various livestock farming situations in France; (iii) to evaluate anti-methane nutritional solutions; (iv) to establish and deploy genomic methane emission assessments, including a synthetic genetic index for 'methane efficiency'; (v) to evaluate the combination of different levers and to quantify methane emissions from various systems; and (vi) to develop a toolkit to support the sector. Around 30 field trials and participatory workshops are planned.
The Methane 2030 project aims to provide all farmers with comprehensive support to help them reduce enteric methane emissions on their farms and continue their efforts to decarbonise. The project brings together 13 scientific and technical partners, as well as 15 dairy and suckler cow farms and experimental sites. These partners include INRAE, APIS-GENE, Idele, les Chambres d’agriculture de Bretagne, ELIANCE, FGE, Races de France and several professional experimental farms from the F@RM XP network. Funding for the project comes from the French government as part of its France 2030 plan and is supported by shareholders through APIS-GENE.
According to the latest figures (Citepa, 2025), GHG emissions in France declined between 2019 and 2023. However, this reduction was below the level required to meet the current carbon budget. All major emitting sectors are participating in GHG reduction, with the livestock sector contributing by reducing methane and nitrous oxide emissions. Efforts aiming to reduce GHG emissions must be stronger to reach carbon neutrality by 2050. Many prospective studies question the role of livestock farming in relation to national low-carbon strategies. However, modelling the potential gains obtained from reducing methane and other GHG emissions in different livestock farming systems, through the combined use of genetic, feeding and management strategies, and their impact, is essential. Experimentation and on-farm evaluation are also necessary to ensure additivity and interaction between different levers are considered.
Overcoming economic and sociological barriers to exploit opportunities in different regions according to their specific strengths and constraints remains challenging. The evolution of livestock farming systems can take several different paths. It is important to discuss the required changes with stakeholders using participatory approaches. Farmers will be remunerated for their efforts if a voluntary sectoral commitment and proactive public policies are in place.
Contribution of the authors
Florence Gondret and Xavier Fernandez contributed more specifically to the introduction, Part 4 and the prospects, and coordinated the writing process. Flavie Tortereau, Maguy Eugène, Donato Andueza, Cécile Martin and Solène Fresco contributed more specifically to Part 1. Simon Roques, Diego Morgavi, Milka Popova, Rafael Muñoz-Tamayo and Valérie Berthelot contributed more specifically to Part 2. Didier Boichard, Pauline Martin and Solène Fresco contributed more specifically to Part 3. Maguy Eugène, Cécile Martin, Amandine Lurette and Mohamed Habibou Assouma contributed more specifically to Parts 4 and 5.
Acknowledgments
This article was first translated with www.DeepL.com/Translator and the authors thank Proof-Reading-Service.com for English language editing. The authors reviewed and edited the content as needed and take full responsibility for the content of the published article.
References
- Andueza, D., Picard, F., Pourrat, J., Vanlierde, A., Nozière, P., Cantalapiedra-Hijar, G., Morgavi, D., De la Torre, A., Dehareng, F., Martin, C., & Renand, G. (2022). Near-infrared spectra from faeces as a proxy of enteric methane emissions and intake in beef cattle [Communication]. 73th EAAP International Symposium on Energy and Protein Metabolism and Nutrition (ISEP). Granada, Spain. https://doi.org/10.1016/j.anscip.2022.07.115
- Archimède, H., Eugène, M., Magdeleine, C. M., Boval, M., Martin, C., Morgavi, D. P., Lecomte, P., & Doreau, M. (2011). Comparison of methane production between C3 and C4 grasses and legumes. Animal Feed Science and Technology, 166, 59-64. https://doi.org/10.1016/j.anifeedsci.2011.04.003
- Assouma, M. H., Hiernaux, P., Lecomte, P., Ickowicz, A., Bernoux, M., & Vayssières, J. (2019). Contrasted seasonal balances in a Sahelian pastoral ecosystem result in a neutral annual carbon balance. Journal of Arid Environments, 162, 62-73. https://doi.org/10.1016/j.jaridenv.2018.11.013
- Assouma, M. H., Lecomte, P., Hiernaux, P., Ickowicz, A., Corniaux, C., Decruyenaere, V., Diarra, A. R., & Vayssières, J. (2018). How to better account for livestock diversity and fodder seasonality in assessing the fodder intake of livestock grazing semi-arid sub-Saharan Africa rangelands. Livestock Science, 216, 16-23. https://doi.org/10.1016/j.livsci.2018.07.002
- Beauchemin, K. A., Ungerfeld, E. M., Abdalla, A. L., Alvarez, C., Arndt, C., Becquet, P., Benchaar, C., Berndt, A., Mauricio, R. M., McAllister, T. A., Oyhantçabal, W., Salami, S. A., Shalloo, L., Sun, Y., Tricarico, J., Uwizeye, A., De Camillis, C., Bernoux, M., Robinson, T., & Kebreab, E. (2022). Invited review: Current enteric methane mitigation options. Journal of Dairy Science, 105(12), 9297-9326. https://doi.org/10.3168/jds.2022-22091
- Bedoya Mazo, S., Posada Ochoa, S. L., Rosero Noguera, R., Bayat, A. R., Blondiaux, P., & Eugène, M. (2023). Quantifying H2 emissions under different nutritional mitigation strategies and its impact on improving the prediction of enteric methane emissions of ruminants. Livestock Science, 277, 105342. https://doi.org/10.1016/j.livsci.2023.105342
- Belanche, A., Hristov, A. N., van Lingen, H. J., Denman, S. E., Kebreab, E., Schwarm, A., Kreuzer, M., Niu, M., Eugène, M., Niderkorn, V., Martin, C., Archimède, H., McGee, M., Reynolds, C. K., Crompton, L. A., Bayat, A. R., Yu, Z., Bannink, A., Dijkstra, J., … Yáñez-Ruiz, D. R. (2023). Prediction of enteric methane emissions by sheep using an intercontinental database. Journal of Cleaner Production, 384, 135523. https://doi.org/10.1016/j.jclepro.2022.135523
- Benaouda, M., Martin, C., Li, X., Kebreab, E., Hristov, A. N., Yu, Z., Yáñez-Ruiz, D. R., Reynolds, C. K., Crompton, L. A., Dijkstra, J., Bannink, A., Schwarm, A., Kreuzer, M., McGee, M., Lund, P., Hellwing, A. L. F., Weisbjerg, M. R., Moate, P. J., Bayat, A. R., … Eugène, M. (2019). Evaluation of the performance of existing mathematical models predicting enteric methane emissions from ruminants: Animal categories and dietary mitigation strategies. Animal Feed Science and Technology, 255, 114207. https://doi.org/10.1016/j.anifeedsci.2019.114207
- Bes, A., Nozière, P., Renand, G., Rochette, Y., Guarnido-Lopez, P., Cantalapiedra-Hijar, G., & Martin, C. (2022). Individual methane emissions (and other gas flows) are repeatable and their relationships with feed efficiency are similar across two contrasting diets in growing bulls. Animal, 16(8), 100583. https://doi.org/10.1016/j.animal.2022.100583
- Blaix, C., Dumont, B., Bloor, J. M. G., Zagaria, C., Fleurance, G., Joly, F., & Huguenin-Elie, O. (2026). Agroecological interventions increase biodiversity and the potential for climate change mitigation in Europe. Agriculture, Ecosystems & Environment, 395, 109938. https://doi.org/10.1016/j.agee.2025.109938
- Blondiaux, P., Cabezas-Garcia, E. H., Senga Kiesse, T., Muñoz-Tamayo, R., Reed, K., & Eugène, M. (2024). Evaluation of the precision and accuracy of models of enteric methane emissions from ruminants using Bayesian inference. Zenodo, 140049835. https://doi.org/10.5281/zenodo.14049835
- Bois, B., Morgavi, D. P., González-García, E., Genestoux, L., Lecomte, P., Ickowicz, A., & Doreau, M. (2020). Indirect measures of methane emissions of Sahelian zebu cattle in West Africa, role of environment and management. Tropical Animal Health and Production, 52(4). 1953-1960. https://doi.org/10.1007/s11250-020-02212-x
- Bougouin, A., Appuhamy, J. A. D. R. N., Ferlay, A., Kebreab, E., Martin, C., Moate, P. J., Benchaar, C., Lund, P., & Eugène, M. (2019). Individual milk fatty acids are potential predictors of enteric methane emissions from dairy cows fed a wide range of diets: Approach by meta-analysis. Journal of Dairy Science, 102(11), 10616-10631. https://doi.org/10.3168/jds.2018-15940
- CGAAER. (2024). Parangonnage sur la diminution des émissions de méthane de l’élevage (Rapport no 23065). https://agriculture.gouv.fr/parangonnage-sur-la-diminution-des-emissions-de-methane-de-lelevage
- Chemineau, P., Peyraud, J.-L., Rieu, M., Magdelaine, P., Duru, M., Vermot-Desroches, C., Allo, C., Brulhet, J., & Le Déaut, J.-Y. (2025). L’élevage intensif en France peut-il être durable ? Quelle conciliation entre producteurs, citoyens et consommateurs ? (Controverses de l’Académie d’Agriculture de France 2024). Académie d’Agriculture de France. https://www.academie-agriculture.fr/publications/publications-academie/avis/rapport-de-lacademie-dagriculture-de-france-lelevage
- Chilliard, Y., Martin, C., Rouel, J., & Doreau, M. (2009). Milk fatty acids in dairy cows fed whole crude linseed, extruded linseed, or linseed oil, and their relationship with methane output. Journal of Dairy Science,92(10), 5199-5211. https://doi.org/10.3168/jds.2009-2375
- Citepa. (2025). . Émissions de gaz à effet de serre et de polluants atmosphériques en France 1990-2024 (Rapport Secten éd. 2025). Citepa. https://www.citepa.org/donnees-air-climat/donnees-gaz-a-effet-de-serre/secten/
- Dall-Orsoletta, A. C., Leurent-Colette, S., Launay, F., Ribeiro-Filho, H. M. N., & Delaby, L. (2019). A quantitative description of the effect of breed, first calving age and feeding strategy on dairy systems enteric methane emission. Livestock Science, 224, 87-95. https://doi.org/10.1016/j.livsci.2019.04.015
- Davoudkhani, M., Rubino, F., Creevey, C. J., Ahvenjä Rvi, S., Bayat, A. R., Tapio, I., Belanche, A., & Muñoz-Tamayo, R. (2024). Integrating microbial abundance time series with fermentation dynamics of the rumen microbiome via mathematical modelling. PLoS One, 19(3), e0298930. https://doi.org/10.1371/journal.pone.0298930
- de Haas, Y., Veerkamp, R. F., de Jong, G., & Aldridge, M. N. (2021). Selective breeding as a mitigation tool for methane emissions from dairy cattle. Animal, 15(Suppl 1), 100294. https://doi.org/10.1016/j.animal.2021.100294
- Doreau, M., Arbre, M., Rochette, Y., Lascoux, C., Eugène, M., & Martin, C. (2018). Comparison of 3 methods for estimating enteric methane and carbon dioxide emission in nonlactating cows. Journal of Animal Science, 96(4), 1559-1569. https://doi.org/10.1093/jas/sky033
- EEA. (2024). National emissions reported to the UNFCCC and to the EU under the Governance Regulation. European Environment Agency. https://doi.org/10.2909/6331f651-8863-4656-a911-669f2a332a1e
- Eugène, M., Sauvant, D., Nozière, P., Viallard, D., Oueslati, K., Lherm, M., Mathias, E., & Doreau, M. (2019). A new Tier 3 method to calculate methane emission inventory for ruminants. Journal of Environmental Management, 231, 982-988. https://doi.org/10.1016/j.jenvman.2018.10.086
- Fakih, I., Got, J., Robles-Rodriguez, C. E., Siegel, A., Forano, E., & Muñoz-Tamayo, R. (2023). Dynamic genome-based metabolic modeling of the predominant cellulolytic rumen bacterium Fibrobacter succinogenes S85. mSystems, 8(3), e01027-22. https://doi.org/10.1128/msystems.01027-22
- FAO. (2023). Pathways towards lower emissions – a global assessment of the greenhouse gas emissions and mitigation options from livestock agrifood systems. Food and Agriculture Organization of the United Nations. https://doi.org/10.4060/cc9029en
- Fischer, A., Edouard, N., & Faverdin, P. (2020). Precision feed restriction improves feed and milk efficiencies and reduces methane emissions of less efficient lactating Holstein cows without impairing their performance. Journal of Dairy Science, 103(5), 4408-4422. https://doi.org/10.3168/jds.2019-17654
- Fresco, S., Boichard, D., Aguerre, S., Fritz, S., & Martin, P. (2025). Short communication: First insights into breeding objectives for reducing methane emissions in Montbéliarde dairy cows. Animal, soumis.
- Fresco, S., Boichard, D., Fritz, S., & Martin, P. (2024a). Genetic parameters for methane production, intensity, and yield predicted from milk mid-infrared spectra throughout lactation in Holstein dairy cows. Journal of Dairy Science, 107(12), 11311-11323. https://doi.org/10.3168/jds.2024-25231
- Fresco, S., Vanlierde, A., Baur, A., Boichard, D., Aguerre, S., Fritz, S., & Martin, P. (2024c). Vers une évaluation génomique des émissions de méthane dans les races bovines laitières françaises [Communication poster]. 27e Rencontres Recherches Ruminants, Paris, France. https://journees3r.fr/textes3r/20240903-vers-une-evaluation-genomique-des-emissions-de-methane-dans-les-races-bovines-laitieres-francaises/
- Fresco, S., Vanlierde, A., Boichard, D., Lefebvre, R., Gaborit, M., Bore, R., Fritz, S., Gengler, N., & Martin, P. (2024b). Combining short-term breath measurements to develop methane prediction equations from cow milk mid-infrared spectra. Animal, 18(7), 101200. https://doi.org/10.1016/j.animal.2024.101200
- Gbenou, G. X., Assouma, M. H., Bastianelli, D., Kiendrebeogo, T., Bonnal, L., Zampaligre, N., Bois, B., Sanogo, S., Sib, O., Martin, C., & Dossa, L. H. (2024b). Enteric methane emissions from zebu cattle are influenced by seasonal variations in rangeland fodder quality and intake. Animal, 18(10), 101320. https://doi.org/10.1016/j.animal.2024.101320
- Gbenou, G. X., Assouma, M. H., Zampaligre, N., Martin, C., Bastianelli, D., Bonnal, L., Kiendrebeogo, T., Sib, O., Bois, B., Sanogo, S., & Dossa, L. H. (2024a). Mesurer, prédire et réduire les émissions de méthane entérique en Afrique subsaharienne. INRAE Productions Animales, 37(1), 7648. https://doi.org/10.20870/productions-animales.2024.37.1.7648
- Goopy, J. P., Woodgate, R., Donaldson, A., Robinson, D. L., & Hegarty, R. S. (2011). Validation of a short-term methane measurement using portable static chambers to estimate daily methane production in sheep. Animal Feed Science and Technology, 166(Special Issue: Greenhouse Gases in Animal Agriculture - Finding a Balance between Food and Emissions), 219-226. https://doi.org/10.1016/j.anifeedsci.2011.04.012
- Guyader, J., Ungerfeld, E. M., & Beauchemin, K. A. (2017). Redirection of metabolic hydrogen by inhibiting methanogenesis in the rumen simulation technique (RUSITEC). Frontiers in Microbiology, 8, 393. https://doi.org/10.3389/fmicb.2017.00393
- Hammond, K. J., Crompton, L. A., Bannink, A., Dijkstra, J., Yanez-Ruiz, D. R., O'Kiely, P., Kebreab, E., Eugène, M. A., Yu, Z., Shingfield, K. J., Schwarm, A., Hristov, A. N., & Reynolds, C. K. (2016). Review of current in vivo measurement techniques for quantifying enteric methane emission from ruminants. Animal Feed Science and Technology, 219, 13-30. https://doi.org/10.1016/j.anifeedsci.2016.05.018
- Huang, R., Romero, P., Belanche, A., Ungerfeld, E. M., Yanez-Ruiz, D., Morgavi, D. P., & Popova, M. (2023). Evaluating the effect of phenolic compounds as hydrogen acceptors when ruminal methanogenesis is inhibited in vitro – Part 1. Dairy cows. Animal, 17(5), 100788. https://doi.org/10.1016/j.animal.2023.100788
- INRA. (2018). Alimentation des ruminants (4e ed.). Éditions Quae. https://hal.science/hal-02789908/
- IPCC. (2019). 2019 Refinement of the 2006 IPCC guidelines for national greenhouse gas inventories. Intergovernmental Panel on Climate Change. In CH4 emissions from enteric fermentation, 4, (pp. 1033–1048, 10.3.). Cambridge University Press. https://www.ipcc.ch/report/2019-refinement-to-the-2006-ipcc-guidelines-for-national-greenhouse-gas-inventories/
- IPCC. (2023). Sections. In Core Writing Team, Lee, H. & Romero, J. (Eds.), Climate Change 2023: Synthesis Report. Contribution of Working Groups I, II and III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. (pp. 35-115). Intergovernmental Panel on Climate Change. https://www.ipcc.ch/report/ar6/syr/downloads/report/IPCC_AR6_SYR_FullVolume.pdf
- Mackie, R. I., Kim, H., Kim, N. K., & Cann, I. (2024). Invited Review – Hydrogen production and hydrogen utilization in the rumen: Key to mitigating enteric methane production. Animal Bioscience, 37(2), 323-336. https://doi.org/10.5713/ab.23.0294
- Martin, C., Morgavi, D. P., & Doreau, M. (2010). Methane mitigation in ruminants: from microbe to the farm scale. Animal, 4(3), 351-365. https://doi.org/10.1017/S1751731109990620
- Martin, C., Niderkorn, V., Maxin, G., Guyader, J., Eugène, M., & Morgavi, D. P. (2021). The use of plant bioactive compounds to reduce greenhouse gas emissions from farmed ruminants. In R. Baines (Ed.), Reducing greenhouse gas emissions from livestock production (pp. 231-260). Burleigh Dodds Science Publishing. https://hal.inrae.fr/hal-03318574
- Martinez-Fernandez, G., Denman, S. E., Walker, N., Kindermann, M., & McSweeney, C. S. (2024). Programming rumen microbiome development in calves with the anti-methanogenic compound 3-NOP. Animal Microbiome, 6(1), 60. https://doi.org/10.1186/s42523-024-00343-2
- Martins, L. F., Cueva, S. F., Wasson, D. E., Almeida, C. V., Eifert, C., de Ondarza, M. B., Tricarico, J. M., & Hristov, A. N. (2024). Effects of dose, dietary nutrient composition, and supplementation period on the efficacy of methane mitigation strategies in dairy cows: A meta-analysis. Journal of Dairy Science, 107(11), 9289-9308. https://doi.org/10.3168/jds.2024-24783
- Meale, S. J., Popova, M., Saro, C., Martin, C., Bernard, A., Lagree, M., Yáñez-Ruiz, D. R., Boudra, H., Duval, S., & Morgavi, D. P. (2021). Early life dietary intervention in dairy calves results in a long-term reduction in methane emissions. Scientific Reports, 11, 3003. https://doi.org/10.1038/s41598-021-82084-9
- Mesgaran, S. D., Baumont, R., Munksgaard, L., Humphries, D., Kennedy, E., Dijkstra, J., Dewhurst, R., Ferguson, H., Terré, M., & Kuhla, B. (2021). Methods in cattle physiology and behaviour research – recommendations from the SmartCow consortium. Publisso. https://books.publisso.de/de/publisso_gold/publishing/books/overview/53/overview%20chapters
- Morgavi, D. P., Cantalapiedra-Hijar, G., Eugène, M., Martin, C., Nozière, P., Popova, M., Ortigues-Marty, I., Muñoz-Tamayo, R., & Ungerfeld, E. M. (2023). Review: Reducing enteric methane emissions improves energy metabolism in livestock: Is the tenet right? Animal, 17(S3), 100830. https://doi.org/10.1016/j.animal.2023.100830
- Morgavi, D. P., Forano, E., Martin, C., & Newbold, C. J. (2010). Microbial ecosystem and methanogenesis in ruminants. Animal, 4(7), 1024-1036. https://doi.org/10.1017/S1751731110000546
- Muñoz-Tamayo, R., Ahvenjärvi, S., Bayat, A. R., & Tapio, I. (2023a). A dynamic mechanistic model of microbial fermentation and methane production in the cow rumen [Communication]. ADSA Annual Meeting, Ottawa, Canada. https://hal.inrae.fr/hal-04181482v1
- Muñoz-Tamayo, R., Chagas, J. C., Ramin, M., & Krizsan, S. J. (2021). Modelling the impact of the macroalgae Asparagopsis taxiformis on rumen microbial fermentation and methane production. Peer Community Journal, 1, e7. https://doi.org/10.24072/PCJOURNAL.11
- Muñoz-Tamayo, R., Davoudkhani, M., Fakih, I., Robles-Rodriguez, C. E., Rubino, F., Creevey, C. J., & Forano, E. (2023b). Review: Towards the next-generation models of the rumen microbiome for enhancing predictive power and guiding sustainable production strategies. Animal, 17, 100984. https://doi.org/10.1016/j.animal.2023.100984
- Muñoz-Tamayo, R., Giger-Reverdin, S., & Sauvant, D. (2016). Mechanistic modelling of in vitro fermentation and methane production by rumen microbiota. Animal Feed Science and Technology, 220, 1‑21. https://doi.org/10.1016/j.anifeedsci.2016.07.005
- Muñoz-Tamayo, R., Popova, M., Tillier, M., Morgavi, D. P., Morel, J.-P., Fonty, G., & Morel-Desrosiers, N. (2019). Hydrogenotrophic methanogens of the mammalian gut: Functionally similar, thermodynamically different - A modelling approach. Plos One, 14(12), e0226243. https://doi.org/10.1371/journal.pone.0226243
- Niderkorn, V., & Jayanegara, A. (2025). Harnessing bioactive-rich forage species in multi-species grasslands: where do we stand? Challenges and future perspectives [Communication]. 23rd EGF symposium, Reading, United Kingdom. https://hal.inrae.fr/hal-05263461v1
- Poux, X., Allen, M., Cain, M., & Patel, V. (2025). Comportement du méthane dans l’atmosphère et analyse des enjeux techniques du PRG100 et du PRG* - application au secteur des ruminants en France et en Europe (Rapport pour l’OFB). AScA. https://hal.science/hal-05033353v1
- Puech, T., & Stark, F. (2023). Diversification of an integrated crop-livestock system: Agroecological and food production assessment at farm scale. Agriculture, Ecosystems & Environment, 344, 108300. https://doi.org/10.1016/j.agee.2022.108300
- Quantin, P., Akinropo, T., Perveeva, K., Hery Ratovoarisoa, M., Assouma, H., & Eugène, M. (2025). Evaluation of the performance of extant models to accurately predict methane emissions from ruminants in a sub-Saharan African context [Communication]. The 9th International Greenhouse Gas & Animal Agriculture Conference, Nairobi, Kenya.
- Ramayo‐Caldas, Y., Zingaretti, L., Popova, M., Estellé, J., Bernard, A., Pons, N., Bellot, P., Mach, N., Rau, A., Roume, H., Perez‐Enciso, M., Faverdin, P., Edouard, N., Ehrlich, D., Morgavi, D. P., & Renand, G. (2020). Identification of rumen microbial biomarkers linked to methane emission in Holstein dairy cows. Journal of Animal Breeding and Genetics, 137(1), 49‑59. https://doi.org/10.1111/jbg.12427
- Romero, P., Huang, R., Jiménez, E., Palma-Hidalgo, J. M., Ungerfeld, E. M., Popova, M., Morgavi, D. P., Belanche, A., & Yáñez-Ruiz, D. R. (2023). Evaluating the effect of phenolic compounds as hydrogen acceptors when ruminal methanogenesis is inhibited in vitro – Part 2. Dairy goats. Animal, 17(5), 100789. https://doi.org/10.1016/j.animal.2023.100789
- Roques, S., Martinez-Fernandez, G., Ramayo-Caldas, Y., Popova, M., Denman, S., Meale, S. J., & Morgavi, D. P. (2024). Recent advances in enteric methane mitigation and the long road to sustainable ruminant production. Annual Review of Animal Biosciences, 12(1), 321-343. https://doi.org/10.1146/annurev-animal-021022-024931
- Saro, C., Martin, C., Cantalapiedra-Hijar, G., Bouchon, M., Chantelauze, C., & Morgavi, D. (2025). Use of 3-nitrooxypropanol in early lactation dairy cows fed a high forage total mixed ration: effect on enteric methane emissions, performance and milk carbon isotopic signature. Journal of Dairy Science, 109(1), 360-371. https://doi.org/10.3168/jds.2025-26843
- Schlecht, E., Hiernaux, P., Achard, F., & Turner, M. D. (2004). Livestock related nutrient budgets within village territories in western Niger. Nutrient Cycling in Agroecosystems, 68(3), 13. https://doi.org/10.1023/B:FRES.0000019453.19364.70
- Serment, A., Giger-Reverdin, S., Schmidely, P., Dhumez, O., Broudiscou, L. P., & Sauvant, D. (2016). In vitro fermentation of total mixed diets differing in concentrate proportion: Relative effects of inocula and substrates. Journal of the Science of Food and Agriculture, 96(1), 160‑168. https://doi.org/10.1002/jsfa.7076
- Thornton, P. K., & Herrero, M. (2010). Potential for reduced methane and carbon dioxide emissions from livestock and pasture management in the tropics. Proceedings of the National Academy of Sciences, 107(46), 19667-19672. https://doi.org/10.1073/pnas.0912890107
- Vanlierde, A., Dehareng, F., Gengler, N., Froidmont, E., McParland, S., Kreuzer, M., Bell, M., Lund, P., Martin, C., Kuhla, B., & Soyeurt, H. (2021). Improving robustness and accuracy of predicted daily methane emissions of dairy cows using milk mid‐infrared spectra. Journal of the Science of Food and Agriculture, 101(8), 3394–3403. https://doi.org/10.1002/jsfa.10969
- Vanlierde, A., Martin, C., Mertens, A., Lorant, N., Le Gall, L., Morel, I., Renand, G., Rochette, Y., Picard, F., Dehareng, F., & Andueza, D. (2024). Estimation of individual CH4 emissions using fecal near infrared spectra for young, dairy and beef cattle [Communication]. 75th Annual Meeting of the European Federation of Animal Science (EAAP), Florence, Italy. https://docs.eaap.org/boa/2024_Florence_EAAP_Book_Abstracts.pdf
- van Lingen, H. J., Fadel, J. G., Yáñez-Ruiz, D. R., Kindermann, M., & Kebreab, E. (2021). Inhibited methanogenesis in the rumen of cattle: Microbial metabolism in response to supplemental 3-nitrooxypropanol and nitrate. Frontiers in Microbiology, 12(2026), 705613. https://doi.org/10.3389/fmicb.2021.705613
- van Lingen, H. J., Niu, M., Kebreab, E., Valadares Filho, S. C., Rooke, J. A., Duthie, C. A., Schwarm, A., Kreuzer, M., Hynd, P. I., Caetano, M., Eugène, M., Martin, C., McGee, M., O’Kiely, P., Hünerberg, M., McAllister, T. A., Berchielli, T. T., Messana, J. D., Peiren, N., … Hristov, A. N. (2019). Prediction of enteric methane production, yield and intensity of beef cattle using an intercontinental database. Agriculture, Ecosystems & Environment, 283, 106575. https://doi.org/10.1016/j.agee.2019.106575
- Vigan, A., Lasseur, J., Benoit, M., Mouillot, F., Eugène, M., Mansard, L., Vigne, M., Lecomte, P., & Dutilly, C. (2017). Evaluating livestock mobility as a strategy for climate change mitigation: Combining models to address the specificities of pastoral systems. Agriculture, Ecosystems & Environment, 242, 89-101. https://doi.org/10.1016/j.agee.2017.03.020
- Yanibada, B., Hohenester, U., Pétéra, M., Canlet, C., Durand, S., Jourdan, F., Boccard, J., Martin, C., Eugène, M., Morgavi, D. P., & Boudra, H. (2020). Inhibition of enteric methanogenesis in dairy cows induces changes in plasma metabolome highlighting metabolic shifts and potential markers of emission. Scientific Reports, 10(1), 15591. https://doi.org/10.1038/s41598-020-72145-w
Abstract
Reducing enteric methane emissions from ruminant livestock is a common goal of many countries to limit global warming. While direct measurements of enteric methane emissions are limited to a small number of animals, indirect predictive methods based on milk or faeces can now be used to assess emissions from large numbers of animals in a variety of contexts. These approaches allow the development of genetic selection models and management practices associated with lower methane emissions for small and large ruminants. A better understanding of the relationships between rumen microbial diversity and the ruminant (host) will help to identify new solutions for modulating rumen microbial populations and hydrogen fluxes, with the aim of reducing methane emissions while maintaining animal production and health. Ruminant diets and herd management are important ways to reduce enteric methane emissions, but the practices are not always suitable for the different pedoclimatic contexts, particularly in hot regions. Trade-off must be evaluated to identify combinations of levers that can reduce enteric methane emissions without compromising the health of animals and the ecosystem services associated with ruminant livestock systems.
Attachments
No supporting information for this articleArticle statistics
 Views: 3279
Views: 3279
Downloads
 PDF: 121
PDF: 121
 XML: 12
XML: 12
Most read articles by the same author(s)
- Asma ZENED, Evelyne FORANO, Céline DELBES, Isabelle VERDIER-METZ, Diego MORGAVI, Milka POPOVA, Yuliaxis RAMAYO-CALDAS, Dominique BERGONIER, Annabelle MEYNADIER, Christel MARIE-ETANCELIN, Ruminants microbiota: state of research and impacts of microbiota on animal performance and health , INRAE Productions Animales: Vol. 33 No. 4 (2020)
- Denis BASTIANELLI, Laurent BONNAL, Philippe BARRE, Serge NABENEZA, Paulo SALGADO, Donato ANDUEZA, Near-infrared spectrometry for the characterization of feed resources , INRAE Productions Animales: Vol. 31 No. 3 (2018): Volume 31: Issue3: Dossier: Feed resources for livestock
- Charlotte DEZETTER, Didier BOICHARD, Nathalie BAREILLE, Bénédicte GRIMARD, Pascale LE MEZEC, Vincent DUCROCQ, Dairy crossbreeding: Pros and cons for Holstein dairy systems , INRAE Productions Animales: Vol. 32 No. 3 (2019)
- Didier BOICHARD, Sandra DOMINIQUE, Marie BÉRODIER, Sébastien FRITZ, Luc DELABY, Corentin FOUÉRÉ, Mekki BOUSSAHA, Anne BARBAT, Use of sexed semen in cattle production , INRAE Productions Animales: Vol. 37 No. 4 (2024)
- Gonzalo CANTALAPIEDRA-HIJAR, Philippe FAVERDIN, Nicolas C FRIGGENS, Pauline MARTIN, Feed Efficiency: towards a better understanding of it as a key element of sustainable livestock systems , INRAE Productions Animales: Vol. 33 No. 4 (2020)
- Christelle KNUDSEN, Cécile BONNEFONT, Laurence FORTUN-LAMOTHE, Karine RICAUD, Xavier FERNANDEZ, Spontaneous liver steatosis in waterfowls: overview on current research and perspectives , INRAE Productions Animales: Vol. 31 No. 2 (2018)
- Nathalie LE FLOC’H, Anne BOUDON, Lucile MONTAGNE, Hélène GILBERT, Florence GONDRET, Bénédicte LEBRET, Louis LEFAUCHEUR, Isabelle LOUVEAU, Élodie MERLOT, Marie-Christine PÈRE, Marie-Christine MEUNIER-SALAÜN, Armelle PRUNIER, Hélène QUESNEL, Welfare and health of gestating sows and growing pigs , INRAE Productions Animales: Vol. 34 No. 3 (2021)
- Christian DUCROT, Maria Belén BARRIO, Alain BOISSY, François CHARRIER, Sergine EVEN, Pierre MORMÈDE, Sandrine PETIT, Marie-Hélène PINARD-VAN DER LAAN, François SCHELCHER, François CASABIANCA, Alain DUCOS, Gilles FOUCRAS, Raphaël GUATTEO, Jean-Louis PEYRAUD, Muriel VAYSSIER-TAUSSAT, Patrick VEYSSET, Nicolas C. FRIGGENS, Xavier FERNANDEZ, Jointly improving animal health and welfare in the transition of livestock systems towards sustainability , INRAE Productions Animales: Vol. 37 No. 3 (2024)
- Marie-Pierre SANCHEZ, Valérie WOLF, Cécile LAITHIER, Mohammed EL JABRI, Éric BEUVIER, Odile ROLET-RÉPÉCAUD, Nicolas GAUDILLIÈRE, Stéphanie MINÉRY, Yuliaxis RAMAYO-CALDAS, Thierry TRIBOUT, Alexis MICHENET, Mekki BOUSSAHA, Sébastien TAUSSAT, Sébastien FRITZ, Agnès DELACROIX-BUCHET, Philippe GROSPERRIN, Mickaël BROCHARD, Didier BOICHARD, Genetic analysis of milk cheese-making traits predicted from mid-infrared spectra in Montbéliarde cows , INRAE Productions Animales: Vol. 32 No. 3 (2019)
- Pascal RAINARD, Gilles FOUCRAS, Didier BOICHARD, Rachel RUPP, Low somatic cell count and susceptibility to mastitis in dairy ruminants , INRAE Productions Animales: Vol. 31 No. 4 (2018)

